
Stoichiometry is essentially an expression of the principle that atoms are conserved in chemical change. The term stoichiometry can refer to any type of calculation that relates the quantities of reactants and products in a chemical reaction. For example, the equation describing the combustion of carbon monoxide to carbon dioxide (Note the need to balance the electric charges)Ī balanced chemical equation expresses the relative number of moles of each component (product or reactant), but because each formula in the equation implies a definite mass of the substance (its molar mass), the equation also implies that certain weight relations exist between the components. Solution: Use the solubility rules table(above) to find the insoluble combinations:ī) Sr 2+( aq) + CO 3 2–( aq) → SrCO 3( s)ĭ) 3 Ca 2+( aq) + 2 PO 4 3–( aq) → 3 Ca 3(PO 4) 2( s)

Write net ionic equations for what happens when aqueous solutions of the following salts are combined: In order to make this determination, it helps to know the solubility rules- which all students of chemistry were at one time required to memorize, but are nowadays usually obtained from tables such as the one shown below.Īlkali metal ions (Li +, Na +, K +, etc.) Predicting the outcome when dissolved salts are mixedįrom the above example involving silver chloride, it is clear that a meaningful net ionic equation can be written only if two ions combine to form an insoluble compound. The formation of a precipitate is often indicated by underscoring. When a solid is formed in a reaction that takes place in solution, it is known as a precipitate. the symbol ( s) indicates that the substance AgCl exists as a solid.the symbol ( aq) signifies that the ions are in aqueous solution, and thus are hydrated, or attached to water molecules.

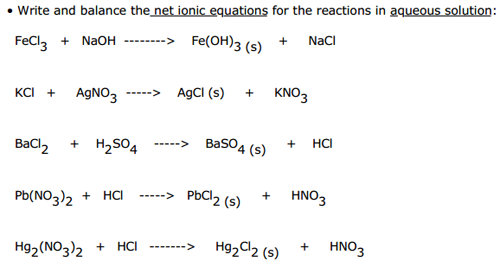
It often happens, however, that we do end up with a fractional coefficient, as in this variant of the above example. Don't worry about this now- just use the appropriate fractional coefficient:įinally, we balance the hydrogens by adding more waters on the right:Īh, but now the oxygens are off again - fixing this also allows us to get rid of the fraction on the left side: In balancing the oxygen, we see that there is no way that an even number of O 2 molecules on the left can yield the uneven number of O atoms shown on the right. It is usually best to begin by balancing compounds containing the least abundant element, so we first balance the equation for carbon: Solution: Begin by writing the unbalanced equation Write a balanced equation for the combustion of propane C 3H 8 in oxygen O 2. Tips and tricks for balancing equations (IsaacsTeach, 7 min) Problem Example 1: combustion of propane Some helpful videos on balancing equations Mark Bishop's excellent online textbook Introduction to Chemistry For more detail, see the guidelines and examples in Chapter 7 of Prof. There is no set “recipe’’ for balancing ordinary chemical equations it is best to begin by carefully studying selected examples such as those given below.
